The purpose of the medical tests is to determine whether a person suffers from osteoporosis, has low bone density and an increased osteoporosis risk, is menopausal and/or has hormonal deficiencies, or an underlying deficiency causing the exacerbation of the bone mass loss.
Tests may be performed to assess the bone density loss, the bone health following a fracture or to monitor the efficiency of the osteoporosis treatment. The imaging diagnosis – a paraclinical investigation – is used to perform osteodensitometry, the main osteoporosis diagnosis test.
Paraclinical investigations:
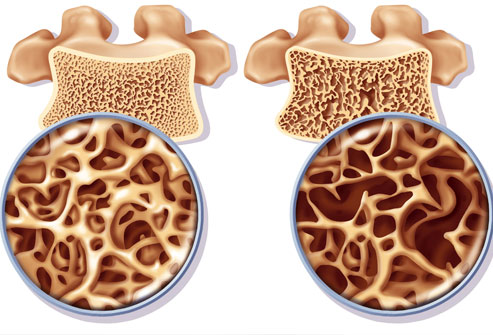
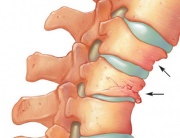
Normal Thoracic and/or Lumbar Spine Radiography (front and lateral view), which may suggest osteoporosis if it highlights an accentuated demineralization at the level of the scanned segments (in which case DEXA is further performed to confirm the diagnosis) sor may indicate an osteoporosis diagnosis if it shows vertebral subsidence in the anterior region of the vertebrae (cuneiform subsidence).
The Bone Mineral Density Test (BMD) ) is the main osteoporosis and low bone mass identification test. One of the preferred and most accurate BMD methods is the dual-energy x-ray absorptiometry (DXA or DEXA). This method uses low intensity X-rays to assess the bone density of the hip and/or of the spine. The BMD value generally is compared to the maximum young adult bone mass value. A BMD value with a standard deviation of one unit below the BMD value of the young adult is regarded as normal. In the case of osteopenia, BMD ranges between -1 and -2.5 standard deviations below the young adult BMD index, while in the case of osteoporosis the BMD values are below -2.5.
Several medical organizations have published osteoporosis screening, guidelines, including the US Preventive Services Task Force. They recommend DXA for the screening of all women above the age of 65 and of women aged between 60 and 64 with fracture risks. The National Osteoporosis Foundation also recommends the screening of men aged above 70, of women aged above 65 and of the middle aged adults falling under the high risk category.
Medical service providers can also use a new instrument, called FRAX, , to more accurately assess the hip, wrist, shoulder or spine fracture risk after the age of 40. Available both online and in hardcopy, the calculator, drafted by the World Health Organization (WHO), takes into account the measured bone density value and other 9 risk factors. The FRAX algorithm assesses the absolute fracture risk. NOF has adapted this algorithm for the USA, and it has been included in the Clinical Guidelines as of February 2008.
Other imaging diagnosis scans that can be performed to measure BDM and identify osteoporosis include the CT scan (computed tomographya) and ultrasounds. A general screening test, called “body composition analysis”, calculates the body mass by assessing the muscle mass, the fat mass, the bone mass and the extracellular water. There are a few different methods to calculate the body composition, including by means of specialized instruments. If the body composition analysis is performed and monitored yearly, it can help identify bone mass losses.
Laboratory tests:
Blood tests may include:
- blood calcium level test – this is a typical osteoporosis test, but it may also be associated to other bone disorders;
- vitamin D test – vitamin D deficit can cause low calcium absorption;
- thyroid function tests – T4 or TSH in order to identify thyroid gland disorders;
- parathyroid hormone test (PTH) – to identify parathyroid gland disease;
- follicle-stimulating hormone (FSH) – menopause identification test;
- testosterone test – to check hormonal deficiencies in men;
- protein electrophoresis – measures the abnormal proteins produced by a certain type of cancer (multiple myeloma), which destroys the bone tissue;
- alkaline phosphatase – measures the high levels that may indicate a bone disorders.
Bone Resorption Assessment:
TBone resorption assessments provide valuable information on the bone mass loss rate. These medical tests can be performed before and after the treatment, in order to check whether the bone mass loss has decreased.
They include:
- c-telopeptide (C-terminal telopeptide of type I collagen (CTx));
- deoxypyridinoline (DPD);
- cross-linked pyridine;
- tartrate-resistant acid phosphatase.
TBone Formation Assessments:
Bone formation assessments provide valuable information on the bone formation rate. Just like bone resorption tests, they may be performed before the treatment and periodically resumed to check the possible bone mass formation rate increase.
They include:
- bone specific alkaline phosphatase (ALP);
- osteocalcin (bone g1a protein);
- P1NP (Procollagen type 1 amino-terminal propeptide).
Below is a presentation of the most widely used bone resorption and formation markers, , determined in blood and/or urine samples. New biomarker researches, which can predict abnormal bone mass losses in various stages of the disease, are ongoing. In the case of most markers, caution is recommended in the interpretation of the results, because of the individual variability associated to the diet, the physical exercise level, and the time of the day when the biological sample was collected.
Blood and urine bone resorption tests include:
C-telopeptide (C-terminal telopeptide of type I collagen (CTx)) – a peptide fragment in the carboxy-terminal region of the protein matrix; helps monitor anti-resorption therapies, such as bisphosphonates or hormone replacement therapy in women during the postmenopausal period and for persons suffering from osteoporosis (low bone mass);
N-telopeptide (N-terminal telopeptide of type I collagen (NTx)) – a peptide fragment in the amino-terminal region of the protein matrix; it is recommended that the test is performed before the osteoporosis treatment and 3-6 months after the therapy;
Deoxypyridinoline (DPD) – is a collagen degradation product, with a ring structure;
Cross-linked pyridine – group of collagen structure degradation products, including DPD; used to monitor the therapeutic response; less specific to bone collagen as than telopeptides;
Tartrate-resistant acid phosphatase (TRAP) 5b – 5b is the TRAP isoform generated by osteoclasts during bone resorption.
Blood and urine bone formation tests include:
Bone specific alkaline phosphatase (ALP) – one of the ALP isoenzymes; it is associated to the functioning of the osteoblasts and it plays a role in bone mineralization; it is recommended that the tests is performed prior to the commencement of the osteoporosis treatment and 3-6 months after the therapy; the results may be influenced by the ALP liver values;
Osteocalcin (bone g1a protein) – a protein exclusively generated by osteoblasts; non-collagen protein of the newly formed bone tissue; it may partially enter the blood flow; the osteocalcin helps predict the bone loss rate amongst postmenopausal women and it may be an indicator of the bone remodelling process rate; it has some degree of usefulness in the choice of the most efficient osteoporosis treatment, but it is less sensitive to changes than telopeptides; it is recommended that the test is performed before the commencement of the osteoporosis treatment and 3-6 months after the therapy. This test may be influenced by the use of Warfarin medicine;
P1NP (Procollagen type 1 amino-terminal propeptide) – generated by osteoblasts; it reflects the bone and collagen formation rates; it may be recommended in parallel with the bone resorption markers, such as C-telopeptide or N-telopeptide; the most sensitive bone formation marker, especially used to monitor bone formation therapies and anti-resorption therapies; this test is recommended prior to the commencement of the osteoporosis treatment and after 3-6 months of therapy.
High blood and urine bone marker levels suggest an increased bone resorption and/or formation rate, but they are unable to suggest the cause. When used to monitor the anti-resorption therapy, the decrease of the bone resorption marker levels indicates a therapeutic response.
If you perform one or several of these tests, you might be advised not to eat before the blood samples are taken. Observe the urine sample collection recommendations, such as taking the sample in the morning, after emptying the urinary bladder.
There are limitations in the clinical usefulness of several bone markers, but researchers continue to study the various ways to expand their usefulness. Their main goal is to standardize the efficiency of the therapies used in the treatment of metabolic disorders and to correctly adjust doses in order to achieve maximum benefits.